MIAMI, FL, Nov. 16, 2022 -- Xtrallux LLC today announced it has received U.S. Food and Drug Administration (FDA) clearance for its photobiomodulating therapy (PBMT) line of wearable hair growth laser caps. This cutting edge product line combines PBMT with wearable technology to provide hair loss patients a convenient, comprehensive way to treat androgenetic alopecia.
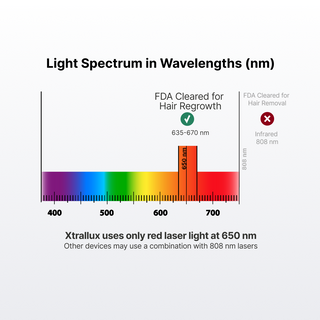
PBMT technology has been used years for years in various medical disciplines. Like its predicates, Xtrallux devices use red laser energy at 650~ nanometers to stimulate hair follicles, the same technology that has been proven to regrow hair in clinical trials. Lasers at this wavelength are healing in nature and have been used by the hair restoration industry for over a decade to restore healthy hair growth from hair follicles.
Androgenetic alopecia is a medical condition through which hair follicles miniaturize over time, eventually ceasing production of hair. This is the most common type of hair loss in men, affecting 50-70% of the male population.1 Women also experience this type of hair loss. It is characterized by distinct patterns of hair loss through which healthy, thick, terminal hairs are replaced by finer, thinner hair which are more sparse and less visible.
"Xtrallux devices provide a safe, effective and convenient method of treating hair loss with professional grade laser strength in a wearable, home-use device," said Carlos Piña, Founder & CEO of the company. "We want to provide the best for our customers."
For full product details on Xtrallux, visit: www.xtrallux.com/products
Indications for Use: The Xtrallux Alpha, Super Plus, Turbo Pro, and Extreme RX are intended to treat Androgenetic Alopecia (AGA) and promote hair growth in males who have Norwood-Hamilton Classifications of Ila to V patterns of hair loss and, females who have Ludwig (Savin) Scale I-1 to I-4, II-1, II-2, or frontal; both with Fitzpatrick Skin Types I to IV. Devices have been cleared for Over-the-Counter use, requiring no prescription from physicians.
About Xtrallux:
Xtrallux LLC is a US-based medical device manufacturer that helps people live happier lives. Our portfolio of laser therapy devices offers various models, providing physicians and patients tools to treat hair loss. Xtrallux aims to serve the needs of people who want to keep their hair looking full and healthy without the need for surgical or medical intervention.
Connect with us at www.xtrallux.com, on LinkedIn at https://www.linkedin.com/company/xtrallux, on Facebook at https://www.facebook.com/xtrallux, on Instagram at @xtrallux and on Twitter @xtrallux
__________________________________
1 https://ishrs.org/patients/types-of-hair-loss/